Filled-in burrows …. or not?
DIGITAL TREASURES
John W.M. Jagt, john.jagt@maastricht.nl
STORIES ON PICTURES
Part 11. Filled-in burrows …. or not?
Isolated scales, bones, teeth and portions of tail, fin and skull, including jaws, of bony fish are not at all rare in the chalks and limestones of Late Cretaceous age in southern Limburg and adjoining areas. However, more or less articulated remains, or even complete fish skeletons, are genuine ‘treasures’. Naturally, this can be linked to the fact that scavengers rapidly did away with dead or moribund fish in the water column or already resting on the sea floor.
Many a collector has already counted him- or herself lucky when coming across an apparently articulated series of scales and bones. This was always possible in the Lanaye Member (Gulpen Formation) and in the lower units of the Maastricht Formation, principally in the Valkenburg and Gronsveld members. I experienced this myself during fieldwork sessions between 1995 en 2019, at the former ENCI quarry, but also elsewhere in the area, as in the Vijlen Member near Haccourt (Liège).
Of note is the fact that this kind of ‘scrapings’ of scales, bones, vertebrae etc. always look like a drawn-out, rather thin burrow (Figure 1). This explains why scientists at the beginning of the nineteenth century considered these to represent eel-like fish.

Figure 1. Lepidenteron lewesiensis (Mantell, 1822) (collection: Frans Smet, St. Gillis-Waas/photograph courtesy of Elena Jagt-Yazykova); ruler in millimetres
On the basis of research elsewhere in Europe (Germany, Czech Republic and Poland), we now know better. These are burrows that were filled in with the sorry remains of rotting fish corpses on the sea floor. What type of animal was responsible for this kind of burrow is a matter of debate; possibly certain types of worm or even crustaceans. Whatever the case may be, these are not ‘body fossils’, but ichnofossils or trace fossils. The name Lepidenteron lewesiensis is available for these burrows.
But – we are not there just yet! There are some tricksters around … The fish illustrated here (Figure 2) is a good example. What is left of this animal (part of the vertebral column, dissociated skull) and the way in which these skeletal elements are ordered, shows this to be a ‘body fossil’ rather than a burrow. In this particular case the fish possibly is a member of the family Dercetidae – elongated, thin fish, of which a number of species has recently been described from the Cretaceous of Liège-Limburg.

Figure 2. A possible representative of the family Dercetidae, with part of the vertebral column and the skull preserved (collection/photograph: Arie de Regt, number AR00161); greatest length 110 mm
This type of ichnofossil constitutes a genuine treasure trove of data on bony fish, because even on the basis of scale type and parts of the skull and jaw fragments, higher-rank groups (families, orders) can be recognised. In short, it is always advisable to take such ‘clad burrows’ home. Whenever they occur in fine-grained chalks, you will have no trouble recovering them, but when they are found in powdery ‘mergel’, it is tricky. What I often did in such cases was collect all in one bag and then, at home, screen wash the material on a sieve (mesh width 0.2 mm), allow the material to dry and then handpick it. The sheer wealth of scales (triangular in Dercetidae) is staggering!
Further reading
Friedman, M., 2012. Ray-finned fishes (Osteichthyes, Actinopterygii) from the type Maastrichtian, the Netherlands and Belgium. In: Jagt, J.W.M., Donovan, S.K. & Jagt-Yazykova, E.A. (eds.). Fossils of the type Maastrichtian (Part 1). Scripta Geologica, 8, 113-142. [download freely: http://www.repository.naturalis.nl/document/455819]
Jagt, J.W.M., 2018. Met visschubben bekleed – het sporenfossiel Lepidenteron lewesiensis uit het Krijt van Luik-Limburg. Gea, 52(1), 15-18.
Jurkowska, A. & Uchman, A., 2013. The trace fossil Lepidenteron lewesiensis (Mantell, 1822) from the Upper Cretaceous of southern Poland. Acta Geologica Polonica, 63, 611-623. [download freely: www.geojournals.pgi.gov.pl/agp/article/view/10371]
Taverne, L. & Goolaerts, S., 2015. The dercetid fishes (Teleostei, Aulopiformes) from the Maastrichtian (Late Cretaceous) of Belgium and The Netherlands. Geologica Belgica, 18, 21-30. [download freely: www.popups.uliege.be/1374-8505/index.php?id=4801&file=1&pid=4794
Strange pits in snail shells
DIGITAL TREASURES
John W.M. Jagt, john.jagt@maastricht.nl
STORIES ON PICTURES
Part 10. Strange pits in snail shells
In the limestone sequences that are exposed in the wider vicinity of Maastricht, concentrations (or enrichments) of shells of snails and bivalves occur at a number of places. Earlier generations of (amateur-) palaeontologists had already noted that and had collected extensively from these levels. Today, opportunities for collecting are few and far between, unfortunately …
If you have ever walked along the beaches of the North Sea, in particular after a southwesterly storm, you will have noted those masses of empty shells, occasionally still articulated and with fleshy remains, arranged in rows: cast onto the beach at high tide and in a wide array of shapes and sizes. Shells assembled in this way belong to animals that, while alive, would rarely (if ever) have encountered one another. They originate from a range of biotopes, each with their own specific features.
In the geobiological literature, this kind of shell beds is to as ‘lumachelle’ or ‘coquina’. However, a differentiation needs to be made between beds of local origin that are not displaced, or moderately so (such as oyster or mussel beds) and concentrations of shells that result from current and storm activity.

Figure 1. The trace fossil Salsariichnus collinsi Donovan, Jagt & Van Knippenberg, 2019, visible on/in the external mould of the snail ‘Cerithium’ tuberculiferum Binkhorst van den Binkhorst, 1861 (collections NHMM PK M2634-1; photograph courtesy of John W. Stroucken); height of shell circa 70 mm
The uppermost 20-50 centimetres of the Meerssen Member in the neighbourhood of the former Curfs quarry (Geulhem, www.limburgs-landschap.nl) are replete with heaps of stacked shells of bivalves, snails and even squid (ammonites). Between 1990 and 2003 a lot of material was collected from this level; on the basis of this, a number of scientific papers have already been published. And, there is more to come …
In only three specimens of the larger-sized snail species (Figures 1-3), all external moulds of the shell (with ornamentation), in the Paul van Knippenberg Collection, the strange, pit-like impressions caught my eye some time ago. I had not seen these before, not from other localities in southern Limburg, nor in other collections!

Figure 2. Salsariichnus collinsi Donovan, Jagt & Van Knippenberg, 2019 in the shell of the snail ‘Cerithium’ tuberculiferum Binkhorst van den Binkhorst, 1861 (collections NHMM PK M3094; photograph courtesy of John W. Stroucken); height of mould circa 40 mm
In order to obtain a better picture of the shape and structure of these pits, latex casts were made. The overall shape and size of the individual pits vary, as does the depth (Figure 2). The casts showed straight away that these pits must have been produced by animals that infested the shell, for whatever reason. For now, it is assumed that this happened only after the death of the snails, as their empty shells lay on the sea floor. In favour of this interpretation is the fact that, more or less, the pits appear to be arranged in rows. In some places they are so close that the circumference and structure of the pits adapted to their ‘neighbour’.
This begs the question what animal was responsible for these pits and what function did they have? If the pits were produced after the death of the snail, there would not have been any advantage (read: food) to the fact that the snail dragged its shell along the sea floor. At times, an empty shell on the sea floor may be in a stable position, but most often these will be at the mercy of currents and/or scavengers that impact the substrate. This means that the shell can roll and lead to the demise of anything attached to the outside.
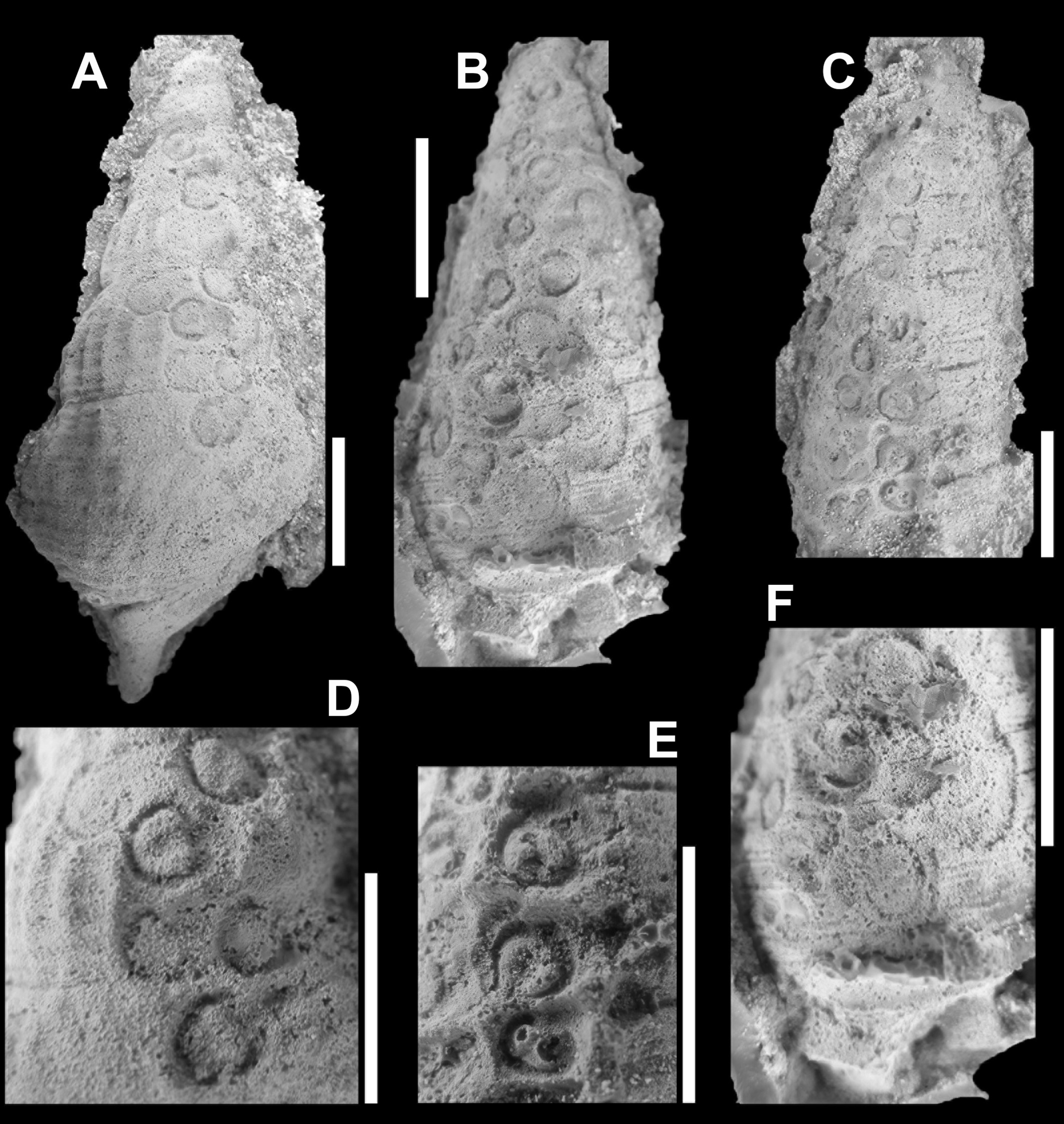
Figure 3. Latex pulls of the ichnofossil Salsariichnus collinsi Donovan, Jagt & Van Knippenberg, 2019 that clearly show that pits in the snail shells consist of a ring with a C-shaped inner margin and a central, deeper-lying pit (from: Donovan et al., 2019, fig. 3; scale bars equal 10 mm)
Be that as it may, this kind of trace deserved to be named, also to facilitate communication about it – the name Salariichnus collinsi was coined for it. Naming traces can be done irrespective of discussions about what kind of animal can be held responsible and why this particular kind of trace has so far only been noted in snail shells.
Further reading
Donovan, S.K., Jagt, J.W.M. & Van Knippenberg, P.H.M., 2019. Clusters of shallow pits in gastropod shells from the type area of the Maastrichtian (Upper Cretaceous, the Netherlands). Bulletin of Geosciences, 94(4), 425-430. [download freely: www.geology.cz/bulletin/contents/art1763]
Jagt, J.W.M., Van Bakel, B.W.M., Cremers, G., Deckers, M.J.M., Dortangs, R.W., Van Es, M., Fraaije, R.H.B,, Kisters, P.J.M., Van Knippenberg, P., Lemmens, H., Nieuwenhuis, E., Severijns, J. & Stroucken, J.W., 2013. Het Vroeg Paleoceen (Danien) van zuidelijk Limburg en aangrenzend gebied – nieuwe fauna’s en nieuwe inzichten. Afzettingen [Werkgroep voor Tertiaire en Kwartaire Geologie], 34(4), 198-230 [download freely: www.natuurtijdschriften.nl/download?type=document&docid=567809]]
Instantaneous death
DIGITAL TREASURES
John W.M. Jagt: john.jagt@maastricht.nl
Natuurhistorisch Museum Maastricht
www.nhmmaastricht.nl
STORIES ON PICTURES
Part 9. Instantaneous death
It will not come as a surprise that in the shallow, subtropical waters (depths of less than 25 metres), periodic storms reeked havoc and led to the sudden death of many an animal on the sea floor. Both direct current energy (impact) and heavy loads of sediment-in-suspension were true killer mechanisms in these settings. The former led to breakage of coral polyps, bryozoan colonies and shells of snails and bivalves, while the latter could smother whole groups of animals on the sea floor by covering them under such a load that escape proved impossible. In geobiological terminology, this phenomenon is known as obrution.
Examples of such obrution have occasionally been found in the limestones of the Maastricht area, but the instance illustrated here ranks amongst the more spectacular ones (Figures 1-3).
A population of >50 individuals, of comparable test size, appears to have been smothered ‘in one go’, as taken by surprise, and killed off instantly.

Figure 1. Gauthieria gr. radiata with spines preserved in a ‘relaxed mode’, but the apical disc is missing, possibly having collapsed into the test (collection/photograph: Mart Deckers, no. NHMM MD 4821); diameter of test around 12 mm
There can be no doubt that the sea urchins were alive when hit by the storm. Their comparable test diameter, and close proximity, suggest that these animals were caught in flagrante delicto … in other words: while reproducing.
With very few exceptions, echinoids shed their egg and sperm cells into the sea water and hope for the best … by huddling close together the chances of generating fertilised eggs (and offspring) increased considerably, of course.

Figure 2. Gauthieria gr. radiata with spines preserved in a ‘relaxed mode’ and apical disc in near-pristine preservation (collection/photograph: Mart Deckers, no. NHMM MD 4821); diameter of test around 12 mm
In all animals that preserve (parts of) their spine canopy, the individual spines are in the ‘relaxed mode’, arranged around the test as the rays of the Sun. When threatened, regular echinoids often group their spines in the direction of the potential attacker – there is no sign of this in the present material. In other words, these animals were really caught by surprise.
The fact that a majority of the individals in this population also preserve (parts of) the lantern, consisting of a large number of isolated plates, teeth and platelets, is another indication of rapid burial and lack of subsequent manipulation by whatever means.

Figure 3. Two tests of Gauthieria gr. radiata with some spines and the lanterns (mouth parts, jaws) preserved (collection/photograph: Mart Deckers, no. NHMM MD 4821); diameter of test around 12 mm
In other words: a genuine snapshot! Instances like this teach us a lot about sea floor conditions, population structures and allow us to determine to what species of echinoid certain dissociated spines and lantern ossicles belong. Without such finds, that would have been guess work.
Further reading
Jagt, J.W.M., 2000. Late Cretaceous-Early Palaeogene echinoderms and the K/T boundary in the southeast Netherlands and northeast Belgium – Part 4: Echinoids. Scripta Geologica, 121, 181-375, 30 pls. [download freely: http://www.repository.naturalis.nl/record/219114]
Jagt, J.W.M. & Deckers, M.J.M. 2012. Het object van de maand. Gauthieria – de hele zee-egel!. Natuurhistorisch Maandblad, 101(3), 52.
Jagt, J.W.M., Van Bakel, B.W.M., Deckers, M.J.M., Donovan, S.K., Fraaije, R.H.B., Jagt-Yazykova, E.A., Laffineur, J., Nieuwenhuis, E. & Thijs, B., 2018. Late Cretaceous echinoderm ‘odds and ends’ from the Low Countries. Contemporary Trends in Geosciences, 7(3), 255-282. [download freely: www.kgs.wnoz.us.edu.pl/ctg/ctg-archive/]
Lazy, yet clever, miniature crustaceans
DIGITAL TREASURES
John W.M. Jagtj: ohn.jagt@maastricht.nl
Natuurhistorisch Museum Maastricht
www.nhmmaastricht.nl
STORIES ON PICTURES
Part 8. Lazy, yet clever, miniature crustaceans
Distant relatives of crabs, shrimp and lobsters (so familiar to us all) include such diminutive to small-sized groups as ostracods and cirripedes, or barnacles. These are well known from the fossil record, also from the wider vicinity of Maastricht.
Less familiar to us amongst barnacles are representatives of the order Acrothoracica. Female individuals of these tiny crustaceans bore into calcareous material (rocks and shelly fossils, such as sea urchin tests, shells of snails and clams) and create a sort of pouch in which they sit. In these pouches, generally comma shaped on the outer surface (entrance) and bag- to kidney shaped in cross section, the females are often accompanied by even tinier males.
In contrast to the majority of other crustaceans, acrothoracicans do not form shells (carapaces) and the only evidence of their former presence left in the fossil record are their typically shaped boreholes. This also means that we can’t assign the producers of these pouches to any biological species because their shell-less corpses rot away following death. Instead, the pouches are named as trace fossils, or ichnofossils. The first name to be introduced in the 1950s for such was Rogerella. Most palaeontologists agree that other names, proposed ater for similar boreholes, should be lumped with Rogerella. I agree.
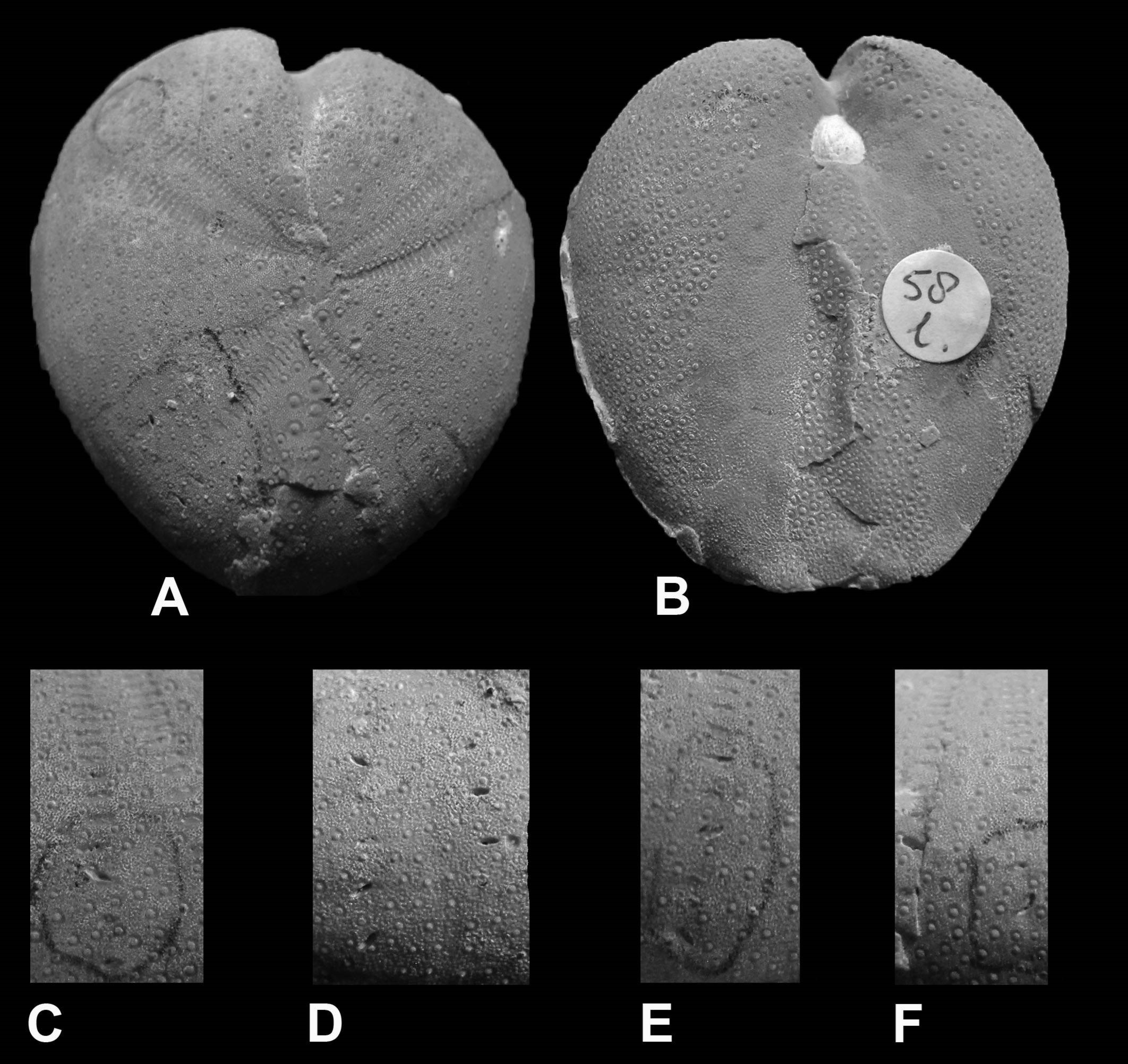
Figure 1. Boreholes in ambulacral pores [lower row] of female acrothoracican crustaceans (ichnogenus Rogerella) in the pores of the sea urchin Cardiaster granulosus [upper row] (collections NHMM EN 58l; photograph: Steve Donovan) (modified from Donovan et al., 2016). The echinoid test measures around 40 mm in length.
To bore holes into a calcareous substrate (of whatever kind) requires time and effort (energy). Then what if the substrate chosen by the crustacean already had pre-existing holes?
The tests of dead sea urchins, when all spines had fallen off, were ideal because their upper surface had five zones of pores. During life, these were the openings for tube feet that served for handling of food particles and respiration (gaseous exchange). Zones with tube feet are called ambulacra; especially close to the highest part of the test, these pores are numerous and close set. Ideal for the lazy so-and-so’s amongst acrothoracicans! Just enter the pore slit and develop the existing hole into a fitting abode without spending too much time and energy (Figure 1).
I now know of such use of ambulacral pores in member of three genera of echinoids, all irregular (Holasteroida), namely Cardiaster, Echinocorys and Hemipneustes, all of Maastrichtian age (69-66.5 million years ago). Only the first named burrowed into the sea floor, so that it would appear the crustaceans bored their holes afte the death of the echinoid.
Let’s all screen the echinoids we have at home or in museum collections – there may be more than meets the eye!
Finally, a word of thanks to Eric Nieuwenhuis and Steve K. Donovan for supply of material and photographs, respectively.
Further reading
Donovan, S.K. & Jagt, J.W.M., 2013. Rogerella isp. infesting the pore pairs of Hemipneustes striatoradiatus (Leske) (Echinoidea: Upper Cretaceous, Belgium). Ichnos, 20, 153-156.
Donovan, S.K., Jagt, J.W.M. & Nieuwenhuis, E., 2016. Site selectivity of the boring Rogerella isp. infesting Cardiaster granulosus (Goldfuss) (Echinoidea) in the type Maastrichtian (Upper Cretaceous, Belgium). Geological Journal, 51, 789-793, doi: 10.1002/gj.2692
Jagt, J.W.M., 2000. Late Cretaceous-Early Palaeogene echinoderms and the K/T boundary in the southeast Netherlands and northeast Belgium – Part 4: Echinoids. Scripta Geologica, 121, 181-375, 30 pls. [download freely: http://www.repository.naturalis.nl/record/219114]
Jagt, J.W.M., Van Bakel, B.W.M., Deckers, M.J.M., Donovan, S.K., Fraaije, R.H.B., Jagt-Yazykova, E.A., Laffineur, J., Nieuwenhuis, E. & Thijs, B., 2018. Late Cretaceous echinoderm ‘odds and ends’ from the Low Countries. Contemporary Trends in Geosciences, 7(3), 255-282. [download freely: www.kgs.wnoz.us.edu.pl/ctg/ctg-archive/]
Adrianna and her fellows – are they sponges?
Adrianna and her fellows – are they sponges?
Dawid Mazurek
If you’re a borer, the muddy, soupy substrate of the vast epicontinental seas during the Late Cretaceous may have seemed the most hostile environment ever. Luckily, there were inoceramids. Big plates of thick, hard shell, their owner resting on the sea floor, almost waiting to be bored (by the borer, that is, not by some dull interlocutor). If you ever venture into one of Opole’s various outcrops of Cretaceous rocks, you will surely encounter inoceramids, and in many cases, these will be covered by sclerobionts (organisms living attached to the skeletons of others, or episkeletozoans), or lined with holes made by borers. The latter may reflect the work of various taxonomic groups; some of the most famous borers living today include bivalves and sponges. One common fossil ichnotaxon (that is, a formally named trace fossil) is Entobia, which consists of an interconnected series of galleries and chambers. If you are ever going to visit Cracow, don’t forget to go and see Entobia cracoviensis borings in situ in the Bonarka nature reserve!

Can you spot the two sun-shaped Entobia borings? Upper Cretaceous of Cracow. Photo by the author.
Sometimes, the arrangement of Entobia may be regular, forming a kind of ‘string of beads’ on the host surface, as visible below.

Cracked and bored inoceramid shell fragment. Turonian of Opole. Photo by the author.
Traditionally, the maker of this kind of structure has been identified as a representative of the demosponge family Clionaidae, but in reality, other sponge groups are likely suspects, too – especially Siphonodictyon, a member of another demosponge family, Phloeodictyidae. Actually, spicules of Siphonodictyon have been recorded (then, as Aka) from Turonian examples of Entobia in England. In the Opole Cretaceous, occasionally stacks of bioeroded, encrusted and heavily fragmented inoceramid shells can be found, and the sediment fill in between can nicely mould the poor state of the shell surface.

Natural moulds of Entobia chambers. Notice that in places, they are so dense that they contact each other. Turonian of Opole. Photo by the author.
But what about Adrianna? This is the nickname of a huge Lewesiceras ammonite, deposited in the University of Opole Museum. Large-sized specimens such as this one are not all that rare in Turonian strata around Opole. What makes it especially peculiar is its pockmarked appearance.

Lewesiceras, nicknamed ‘Adrianna’ from Opole. Exhibition at the University of Opole Museum. Photo by E.Yazykova
If you compare the last two specimens illustrated above, you might wish to consider an unpleasant explanation for Adrianna’s ‘acne’. The ammonite shell was heavily infested by boring sponges. Luckily, this was post mortem, meaning after the ammonite had died and sunk to the sea floor. Similarly bored nautiloids have recently been published by Polish palaeontologists. I got convinced! What about you, dear reader? Adrianna’s ‘face’ remains a wee bit mysterious; maybe there are alternative explanations out there? Somewhere ….
Additional reading:
Bromley, R.G. & Schönberg, C.H.L. 2008. Borings, bodies and ghosts: spicules of the endolithic sponge Aka akis sp. nov. within the boring Entobia cretacea, Cretaceous, England. In: Wisshak, M. & Tapanila, L. (Eds.), Current Developments in Bioerosion. Springer-Verlag: Berlin/Heidelberg, pp. 235–248.
Bromley, R.G., Kędzierski, M., Kołodziej, B. & Uchman, A. 2009. Large chambered sponge borings on a Late Cretaceous abrasion platform at Kraków, Poland. Cretaceous Research 30 (1): 149–160.
Malchyk, O., Łukowiak, M. & Machalski, M. 2017. Sponge borings on the Late Cretaceous nautilids from Poland and Western Ukraine: taphonomic implications. 8th International Meeting on Taphonomy and Fossilization, 14-17 September 2017, University of Vienna, Austria.
hidden and well armed
DIGITAL TREASURES
John W.M. Jagt: john.jagt@maastricht.nl
Natuurhistorisch Museum Maastricht
www.nhmmaastricht.nl
STORIES ON PICTURES
Part 7. Both hidden and well armed
The highest 10 metres (or so) of the limestone sequence of Sint-Pietersberg, south of Maastricht, document deposition of material in a shallow-water (less than 25 metres depth), subtropical setting that would have looked closely similar to snorkling grounds in the Caribbean, the Indonesian archipelago and the Great Barrier Reef in Australia.
In such settings, with numerous corals and other colonial organisms that attached directly to the sea floor, there would have been lots of microhabitats. These were mostly put to good use by light-shy (bright sunlight) animals that were mostly firmly (and permanently) fixed to hard substrates. Favourite places to settle on, away from the hustle and bustle of the sea floor, were the walls of burrows excavated into the sea floor by certain groups of decapod crustaceans, such as mud shrimps (Callianassoidea). These burrows had widely diverging diameters, which also determines the size of the animals that entered them to live inside.
Within these burrows, there was of course limited space and food availability, meaning staunch competition for resources! A true ‘arm race’ is seen amongst several species of bryozoans and tube worms on the walls of such vacated crustacean abodes.
Brachiopods, a special group of invertebrates closely related to bryozoans, include mostly members of the family Craniidae. Superficially these shells resemble a human skull (or ‘cranium’ in Latin), because the muscle pads look like eyes and the ridge between them likens a nose. The lower part then, down to the margin of the shell, would be the (smiling) mouth …
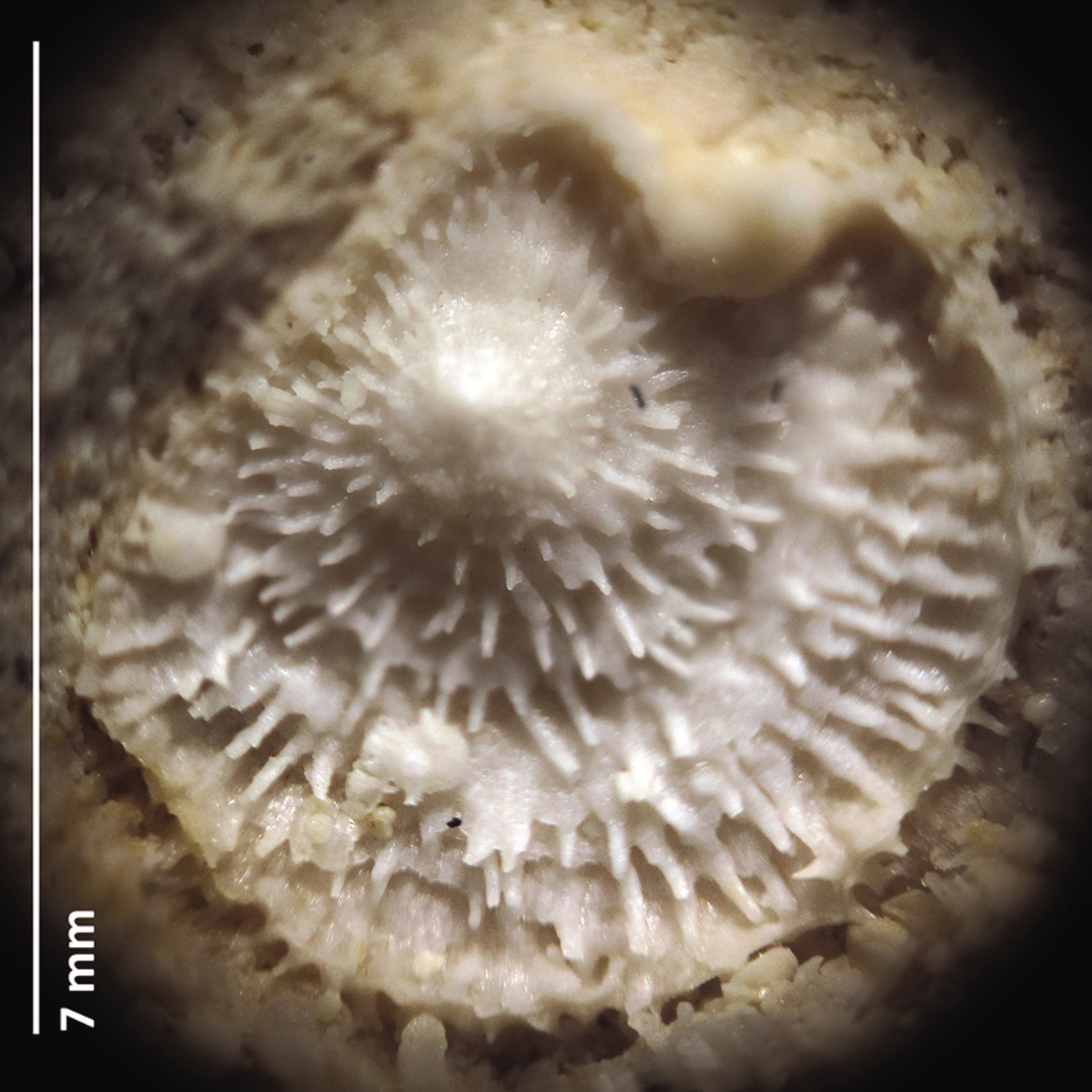
Figure 1. Ancistrocrania cf. comosa, a complete individual with both valves still articulated; Meerssen Member, former ENCI quarry (collection/photograph: Oliver Kesselhut) (see also Kesselhut & Jagt, 2016)
After the death of the animal, the muscles quickly started rotting away and the two halves of the shell separated and got dispersed. Rarely these are preserved as in the example illustrated here (Figure 1).
It would seem that brachiopods did not make for good food during the Late Cretaceous, but still … a predator might have been tempted. Being permanently fixed to a substrate also means that there is no way of escaping attack. The only way is to become less palatable and/or carry armature (spines; Figure 2). Certainly, the present brachiopod would have looked sufficiently intimidating to shy away from!

Figure 2. Ancistrocrania cf. comosa; note the various growth phases [from the top to the margin of the shell] and the variable length of the spines; the valve fixed to the substrate can be seen near the lower margin of the photograph (collection/photograph: Oliver Kesselhut) (see also Kesselhut & Jagt, 2016)
Further reading
Bosquet, J., 1854. Nouveaux brachiopodes du système Maestrichtien. Verhandelingen van de Commissie voor de Geologische Kaart en Beschrijving van Nederland, 2, 197-203.
Bosquet, J., 1859. Monographie des brachiopodes fossiles du terrain Crétacé supérieur du Duché de Limbourg. Première partie. Craniadae et Terebratulidae (subfamilia Thecidiidae). Mémoires pour servir à la description géologique de la Néerlande, 3, 1-50.
Kesselhut, O. & Jagt, J.W.M., 2016. Een vastzittende armpotige met lange stekels. Ancistrocrania cf. comosa uit het Maastrichtse Krijt. Gea, 49(3), 68-69 [www.natuurtijdschriften.nl]
Kruytzer, E.M., 1969. Le genre Crania du Crétacé supérieur et du Post-Maastrichtien de la province de Limbourg néerlandais (Brachiopoda, Inarticulata). Publicaties van het Natuurhistorisch Genootschap in Limburg, 19(3), 4-42.
Digging tunnels
DIGITAL TREASURES
John W.M. Jagt
Natuurhistorisch Museum Maastricht
www.nhmmaastricht.nl
STORIES ON PICTURES
Part 6. Digging tunnels
In many places, the sea floor in our region during the Late Cretaceous (69-66 million years ago) was littered with empty snail shells, loose clam valves, empty sea urchin tests and more of such potential substrates for other animals to attach to. But, there were other organisms lying in wait to tap into those reserves – animals that, for whatever reason (hiding, food), bore into a calcareous substrate. By doing so, these animals leave behind traces. Those traces can be lumped and referred to as bioerosion and they can, provided that they can be recognised easily and are characteristic, even be formally named (Latin name), as an ichnofossil taxon.
Ichnofossils are of great importance for the reconstruction of ancient environments and sea floor conditions. In view of that it is a real pity that their study in the Maastricht area has started in earnest only recently – meanwhile we have embarked on numerous projects that will allow us to catch up.
In those cases where the boring was subsequently filled with silica, which has later ‘crystallised’ into flint, details of the boring are well preserved. So well even that the boring illustrated here (Figure 1), in the loose valve of a brachiopod, could be linked to an ichnofossil that is around 75 million years older and was known only from the Upper Jurassic (Tithonian) in southern England and the Paris Basin (France).

Figure 1. Flint-infilled boring assigned to Cunctichnus probans; scale bar is 10 mm (collections NHMM EN 57t, donated by Eric Nieuwenhuis) (photograph courtesy of Steve Donovan)
The species name of the ichnofossil, probans, is very apt! Why? Well, it can be clearly seen that the animal responsible for the boring (a worm?) did a test, every time it turned a corner, and that up to three times (‘just testing …’), in order to see what the next move needed to be. This testing resulted in up to three ‘digits’, of comparable length, in those places where there was a curve in the boring (Figure 1).
The fact that this ichnofossil has now been found at two widely separated localities, and with 75 million years in between, shows once again that certain species of ichnofossils can only be recognised if the substrate is similar and if they are actively searched for.
Here too there are ample chances for future discoveries …
Further reading
Donovan, S.K., Jagt, J.W.M. & Nieuwenhuis, E.A.P.M., 2015. The boring Cunctichnus probans Fürsich, Palmer and Goodyear, 1994, from the type Maastrichtian (Upper Cretaceous, northeast Belgium). Ichnos, 22(1), 19-21, http://dx.doi.org/10.1080/10420940.2014.988211
Boring mussels, or rather not?
DIGITAL TREASURES
John W.M. Jagt
Natuurhistorisch Museum Maastricht
www.nhmmaastricht.nl
STORIES ON PICTURES
Part 5. Boring mussels, or rather not?
Ask anyone in the street to name a few molluscs and you can rest assured that will hear ‘mussels’ … Whether you like them as food or not; everyone has a mental image of mussels. Dark blue to black shells, with a light purplish blue, mother-of-pearl inside. The ligament that connects the valves doesn’t amount to much. That’s about it …. all in all, quite boring shells. They are attached to any kind of substrate by means of strong, hairlike threads, the byssus.
All of the above doesn’t wash with fossil mussels from limestones in southern Limburg and neighbouring areas. Occasionally, silicified shells are found, either as isolated valves or still articulated, and only rarely are their shells smooth and lack ornament.
If you really pay attention, there are lots of varieties where mussels are concerned, one even more appealing than the other. They do deserve better, although most collectors of fossils ignore them most of the time.
Here is a random selection; I could have added many more. Have a look yourself and google these names: Septifer, Inoperna or Crenella. In general, these are shells of modest size, but there are giants that reach lengths of 80 mm. Very small species, thin shelled and thus fragile, can sometimes be found inside small borings or crevices in hardened limestone beds, in numerous specimens that are arranged as roof tiles.
In overall shell shape, almost all mussels are more or less similar. It is the growth lines, growth halts (thicker ridges) and ornament that allow them to be differentiated. The specimen in Figure 1 has numerous thin ribs that extend from the top to the margin, and where crossing the growth lines, form small granules, as a kind of fragile pearl necklace.
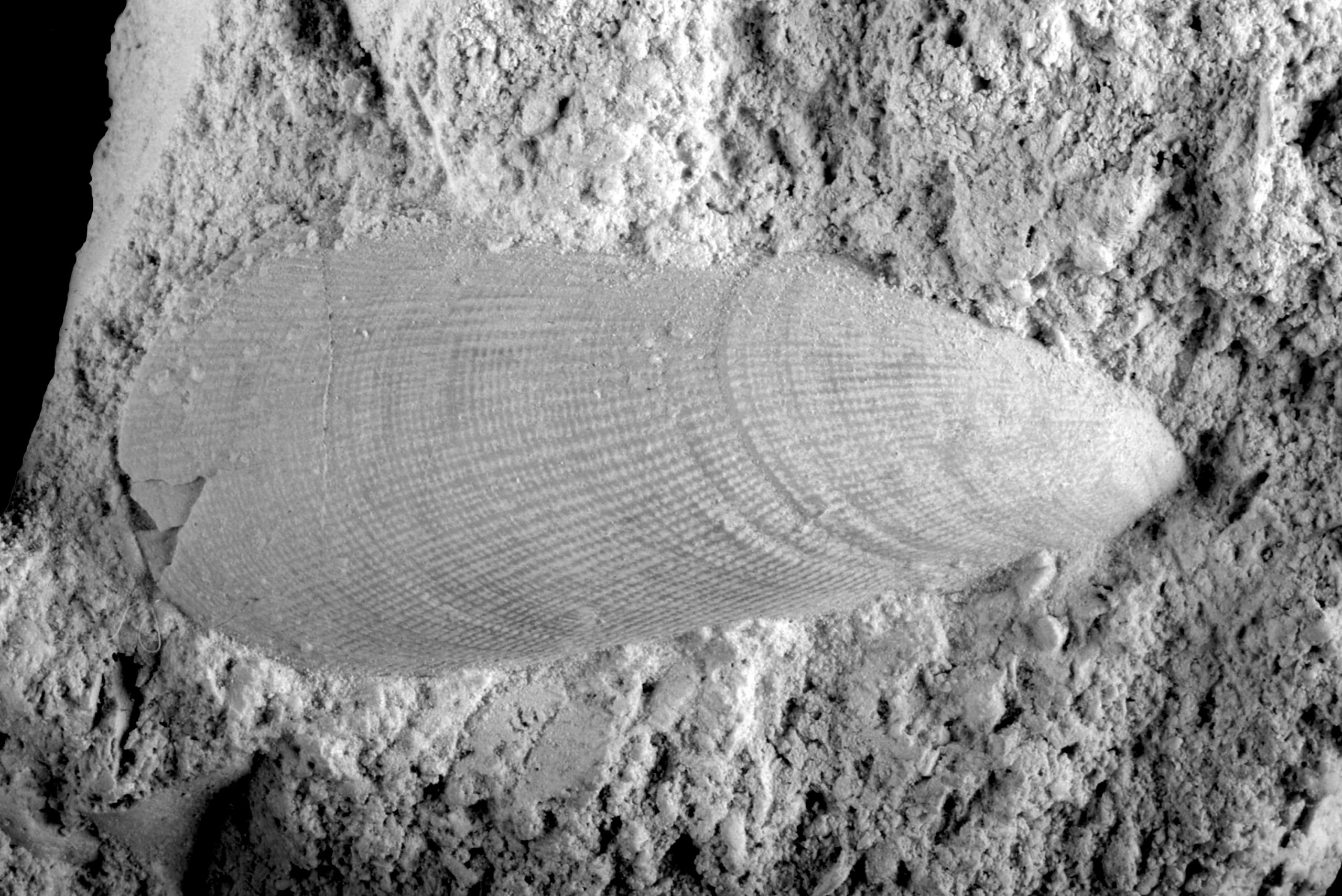
Figure 1. Brachidontes sp. (collections NHMM JJ 14965) (photograph courtesy of Wilfried Miseur). Natural size 32 mm

Figure 2. Brachidontes sp. (W. van Rijsselt Collection) (photograph courtesy of John W. Stroucken). Scale bar in millimetres
Of that type a few species appear to be represented; these differ from each other in details of shell ornament. The specimen in Figure 2 has conspicuous growth halts, at more or less regular distances.
A different type has V-shaped (chevron) ribs, with the base of the V directed towards the top of the shell (Figure 3). Of this type also a number of species are known (Figure 4), but it is also possible that these two shells belong to one and the same species but represent a different ontogenetic stage: juvenile, subadult and adult. To be able to determine this, more material is needed.

Figure 3. Lecompteus sp. (collections NHMM JJ 11526) (photograph courtesy of Wilfried Miseur). Natural size 30 mm

Figure 4. Lecompteus aff. radiatulus (W. van Rijsselt Collection) (photograph courtesy of John W. Stroucken). Scale bar in millimetres
Field work during the last twenty years has demonstrated that mussel banks never occurred during the Late Cretaceous in the vicinity of Maastricht, in contrast to oyster banks. However, Septifer was very common at some levels within the Meerssen Member (subunits IVf-5 and -6), as has been noted at the former ENCI quarry. Of note is also that fossil mussels seen in the field, or collected, very rarely reveal (healed) predation traces. This is remarkable, because, being byssally attached molluscs, mussels could barely defend themselves against attacking predators. The same holds true for oysters, but those often show healed shell fractures. In short: lots of questions remain where the theme of Cretaceous mussels is concerned.
Further reading
Holzapfel, E., 1889. Die Mollusken der Aachener Kreide. II. Lamellibranchiata. Palaeontographica, 35, 139-268.
Jagt, J.W.M., Van Rijsselt, E. & Van Rijsselt, W., 2014. Opmerkelijke Luiks-Limburgse Krijtfossielen. Deel 21. Stokoude mosselen. Natuurhistorisch Maandblad, 103(7), 190-193.
Van de Poel, L., 1959. Faune malacologique du Hervien. Troisième note (première partie). Bulletin
de l’Institut royal des Sciences naturelles de Belgique, 35(15), 1-28.
Vogel, F., 1895. Beiträge zur Kenntniss der Holländischen Kreide. I. Lamellibranchiaten aus der Oberen Mucronatenkreide von Holländisch Limburg. II. Die Fossilien des Neocomsandsteins von
Losser und Gildehaus. Sammlungen des Geologischen Reichsmuseums in Leiden, neue Folge, 2, 1-64.
Malformed shark teeth
DIGITAL TREASURES
John W.M. Jagt
Natuurhistorisch Museum Maastricht
www.nhmmaastricht.nl
STORIES ON PICTURES
Part 4. Malformed shark teeth
In addition to their streamlined, torpedo-shaped body with notable belly, dorsal and caudal fins, sharks have another typical feature in common: a so-called revolver dentition. Whenever they bite into prey, teeth in their upper and lower jaws are lost; how many per bit varies widely on the prey taken. To prevent their marvelllous jaws from becoming inefficient, there is a constant supply of teeth from ‘below’. Immediately behind and underneath, i.e., into the mouth, the functional teeth in the upper and lower jaw, the new generations of teeth are lying in wait. These then shift forwards and pop into position whenever needed.
This wasn’t any different during the Late Cretaceous, more than 66 million years ago. In view of the fact that sharks (and their cousins, the rays) have a cartilagenous skeleton, isolated teeth generally are the sole remains of sharks that fossilise. Also in our area. Of the large majority of the 100+ fossil species of shark and ray in the environs of Maastricht only teeth are known to us. Ever since the early days of fossil hunting in the area, at the end of the eighteenth century, they have been illustrated.
A sought-after prize to collectors of fossils are teeth of a large-sized shark of the extinct family, Anacoracidae: Squalicorax pristodontus (Figures 1-3). Often with the enamel of a beautifully light to dark blue colour, with typical serrated edges and a large, flattened root. Naturally, collectors favour completely preserved teeth, but, to be brutally honest, malformed teeth are much more interesting … A couple of examples of such ‘diseased’ teeth from the Maastricht Formation in Limburg and neighbouring Belgian territory are illustrated here.

Figure 1. Lateral tooth of Squalicorax pristodontus (Sjeng Smits Collection; photographs courtesy of Mart Deckers)
As noted above, the nature and resilience of the prey item that the shark sinks its teeth into determines the nature of the damage to those teeth and the loss thereof. Broken tooth crowns can be easily distinguished. But, there are also teeth that have ‘a flaw’, because they are crooked, have extra serrated edges near the root (or a double row of such along te tooth cutting edge) or a malformation in the enamel. This kind of pathologies often is linked with genetic deficiencies during tooth growth. A ‘twinned’ main cusp has also been found (Figure 4). This looks a bit like a Siamese twin; two directly adjacent teeth become fused, for whatever reason.

Figure 2. Lateral tooth of Squalicorax pristodontus with an additional enamel comb just above the root margin (collection/photographs courtesy of Math van Es)

Figure 3. Anterior tooth of Squalicorax pristodontus with a faint subdivision of the crown and an outgrowth of the enamel on the external tooth surface (collection/photographs courtesy of Math van Es)

Figure 4. ‘Twinned cusp’ of a mako, Carcharias sp.; a fusion of two adjacent teeth (collection/photographs courtesy of Math van Es)
This kind of ‘diseased teeth’ allows us to look into the faces of these extinct sharks, so to speak. The chances that we shall ever find anything else of their cartilaginous skeleton are very slim, indeed, so we need to make do with their loose teeth. This makes these animals even more interesting and allows us a glimpse of the life in this shallow and warm sea that was teeming with life.
Further reading
Bor, T., 2013. Terminologie en determinatie van haaien- en roggentanden. Afzettingen (Werkgroep
voor Tertiaire en Kwartaire Geologie), 34(4), 116-137. [in Dutch; download freel: www.natuurtijdschriften.nl]
Brignon, A., 2015. Faujas de Saint-Fond, Reinwardt, Cuvier et les poissons fossiles du Crétacé
de la «Montagne Saint-Pierre» de Maastricht (Pays-Bas). Geodiversitas, 37(1), 59-77. [download freely: www.sciencepress.mnhn.fr/en/periodiques/geodiversitas/37/1]
Herman, J., 1977. Les sélaciens des terrains néocrétacées & paléocènes de Belgique & des contrées limitrophes. Eléments d’une biostratigraphie intercontinentale. Mémoires pour servir à l’explication des cartes géologiques et minières de la Belgique, 15 (1975), 5-401.
Jagt, J.W.M., Deckers, M., Van Es, M., Severijns, J. & Smits, S., 2018. Opmerkelijke Luiks-Limburgse Krijtfossielen. Deel 34. Misvormde haaientanden. Natuurhistorisch Maandblad, 107(12), 251-254.
A lucky strike
John W.M. Jagt
DIGITAL TREASURES
STORIES ON PICTURES
Part 3. A lucky strike
Amongst the numerous species of bivalves, such as oysters, mussels, cockles, scallops and others, a twofold division can be made. On the one end there are species with a shell consisting of an unstable form of calcite, aragonite, and on the other, those with a calcitic shell. During fossilisation, aragonitic shells dissolve relatively easily, unless they are transformed into calcite or silica during the early stages of fossilisation. This happens under certain conditions. However, of aragonitic shells we usually find solely the infill of the inner surface of the shell, the so-called internal mould (or steinkern) and an external mould of the outer surface of the shell, with or without ornament or sculpture.
In such cases, the use of silicone rubber is called for, because an artificial shell can be produced in this way. This will often reveal details of the hinge and ornament that are of importance for identifying the shells to genus or species. A large number of species in the Maastricht limestones of Late Cretaceous age are known only as internal and external moulds, which is really frustrating, to say the least. But – we can learn a lot from studying such fossils. In addition, it means that the list of species not previously recorded from the type area of the Maastrichtian (i.e., the vicinity of Maastricht), is steadily growing.
Rare are bivalves that have a combined aragonitic and calcitic shell. When, following the animal’s death, the shells disarticulate after the rotting away of the ligament fibres that keep the valves together, they can be covered by sediment on the sea floor. Then the aragonite dissolves, creating a kind of vacuum. This is then, sometimes, filled in by sediment, but not always. And precisely this kind of fossils gets us fooled every time (Figure 1A, B)!


Figure 1A, B. Pulvinites elenae, the type specimen (collections NHMM JJ 14601a, b) (photographs courtesy of Allart van Viersen). A is the sediment infill (steinkern) of the shell, with imprints of musckes and ligament with ‘teeth’; B is the shell proper, seen from the inside. Natural width 20.5 mm
Superficially, the calcitic shell (Figure 1B) does resemble an oyster, with typical, close-set, faintly irregular growth lines. But, not a trace of imprints of muscles in sight, nor of a ligament groove in the hinge. In short: not one step closer to revealing the identity of this shell – put aside for a bit, until …
… the moment when a piece of indurated limestone is smashed in two at the former ENCI quarry and the fossil here illustrated is staring me in the face. Eureka! Now it is clear what this is – the internal mould (Figure 1A) shows the ligament groove (with ‘teeth’) and weak imprints of muscles (in low-angle light), while the associated calcitic shell (Figure 1B) lacks any trace of these. This means that another shell layer must have been present originally; an aragonitic one that has fallen victim to dissolution at an early stage.
Finally this bivalve could be named and added to the growing list of species on record from the Maastrichtian. It is named after my favourite palaeontologist …
Further reading
Jagt, J.W.M., 2018. Latest Cretaceous Pulvinites (Bivalvia, Pterioidea, Pulvinitidae), new to the Maastrichtian type area, The Netherlands. Basteria, 82(4-6), 103-109.
A sea urchin that suffered a lot ...
John W.M. Jagt
DIGITAL TREASURES
stories on pictures
Part 2. A sea urchin that suffered a lot …
It was Professor Umbgrove who, in the mid-1950s, observed that all the streets of the city of Maastricht would have been cobbled with the tests of the sea urchin Hemipneustes striatoradiatus (Leske, 1778) if that species had been even more successful in reproducing … Naturally, that is a bit of an exaggeration, but still – this echinoid is very well represented in all collections of Cretaceous fossils, at museums, universities and private collectors alike.
The large-sized (up to 11 cm, or more), bulbous test, with flattened lower side and large openings for mouth and anus on that side, cannot be overlooked. It is a much desired collectors’ item. Just because this echinoid has a bulbous outline, we know that the animal could only burrow superficially into the sea floor. The largest part of the test remained visible on the sea floor, as the animal ploughed through it in search of food particles.
To be well visible on the sea floor also means that you are spotted by predatory animals, such as fish, sharks, rays and crustaceans (crabs, lobsters). Quite a number of specimens of Hemipneustes shows healed scars on their tests – silent witnesses of predator bites that the echinoid was able to repair. A times such attacks led to malformation or growth disturbance of tests, as seen in Figures 1 and 2. These are clear indications of external influence. But, also internally things could go horribly wrong from time to time as a result of disease and/or infections. The various collections screened by me contain quite a number of interesting examples of such. These will be briefly discussed on a later occasion.

Figure 1. Malformation in Hemipneustes striatoradiatus (collections of the Natuurhistorisch Museum Maastricht, MK 860 A2; photographs courtesy of Mart Deckers)

Figure 2. Notable shell damage, repair and malformation in Hemipneustes striatoradiatus (collections of the Natuurhistorisch Museum Maastricht, MK 890 A8; photographs courtesy of Mart Deckers)
Even after the echinoid’s death, when all spines had become dissociated and scattered, the test would remain available on the seafloor to other animals to attach to or bore into. In Figure 3 a spectacular instance of a bored test is illustrated. The hole penetrates the echinoid test completely, from top to bottom; the diameter of the hole increases slightly, as can be seen in the photograph. The shape of the hole suggests that it was made by a boring bivalve (mollusc) of the family Lithophagidae (literally: stone eaters). That bivalve must have made a small mistake, because normally boring bivalves remain lodged in their own boreholes until they die. Such molluscs often have a combination of mechanical and chemical tricks up their sleeve while boring into calcareous material. The coasts in countries bordering the Mediterranean Sea sport great examples of this.

Figure 3. Hemipneustes striatoradiatus with boring of a bivalve (Jules Snellings Collection; photographs courtesy of Mart Deckers)
Further reading
Jagt, J.W.M., 2000. Late Cretaceous-Early Palaeogene echinoderms and the K/T boundary in the southeast Netherlands and northeast Belgium – Part 4: Echinoids. Scripta Geologica, 121, 181-375, 30 pls. [download freely: http://www.repository.naturalis.nl/record/219114]
Jagt, J.W.M., Van Bakel, B.W.M., Deckers, M.J.M., Donovan, S.K., Fraaije, R.H.B., Jagt-Yazykova, E.A., Laffineur, J., Nieuwenhuis, E. & Thijs, B., 2018. Late Cretaceous echinoderm ‘odds and ends’ from the Low Countries. Contemporary Trends in Geosciences, 7(3), 255-282. [download freely: www.kgs.wnoz.us.edu.pl/ctg/ctg-archive/]
Umbgrove, J.H.F., 1956. Ons land zeventig millioen jaar geleden. Levensschetsen uit de Krijtperiode, xiii + 150 pp., ’s Gravenhage, Martinus Nijhoff.
Where have all those droppings of our mosasaurs gone?
John W.M. Jagt
Natuurhistorisch Museum Maastricht
www.nhmmaastricht.nl
STORIES ON PICTURES
Part 1. Where have all those droppings of our mosasaurs gone?
That certainly is a question that is on the mind of many a museum visitor, but only the braver ones dare ask … That is, perhaps, also a good thing, because the answer to that question is not at all simple.
It remains a mystery. To date, we have documented at least six species of mosasaur from the chalk deposits in Limburg and neighbouring areas. Of these isolated tooth crowns are found, but also portions of their skeleton and, very rarely, articulated skeletal remains. During the last twenty years we have had absolutely no reason for complaint – between 1998 and 2015 we could recover the remains of four individuals at the former ENCI quarry (Sint-Pietersberg, Maastricht). Those mosasaurs, nicknamed Bèr, Kristine, Carlo and Lars, belong to the species Prognathodon saturator, Plioplatecarpus marshi, Prognathodon cf. sectorius and Mosasaurus hoffmannii, respectively. Skeletal remains of the last-named species are the commonest.
The larger-sized mosasaur species in particular were apex predators – they occupied the top positions in the food chain and will have feasted on many a fish, shark, turtle and other mosasaur. To eat that much also means, by definition, to produce quite an amount of poo … Then where are those droppings?
We do know fossilised shark poo of species that lived in the same area and at the same time, also from Sint-Pietersberg (figure 1). They are not that big, often of a spiral nature and have a layered appearance. This is directly connected to the structure of the shark intestines. To learn more about this, reference is made to a paper by Hunt et al. (2015) that can be downloaded freely.

Figure 1. Fossilised shark poo from limestones at Sint-Pietersberg (Maastricht), with typical layers arranged in a sort of spiral (photograph/collection: Mart Deckers, number MD 3948c)
Mosasaurs were much larger than sharks, and would have therefore have produced larger droppings. And yet, we haven’t been able to find these, not even in association with the partial skeletons of Bèr, Carlo and Lars. That is positively weird. It becomes even weirder when we realise that fossilised mosasaur poo, or coprolites, has already been recorded from North America.
Very recently, even a resounding name has become available for mosasaur dropping (figs. 2, 3); really! It is Beechybromus wellschi – it was introduced by Hunt & Lucas (2018). This kind of coprolites has been found in Kansas (USA) and Saskatchewan (Canada) in direct proximity of skeletal elements of mosasaurs. With that in mind, it would appear logical to interpret these as mosasaur droppings.
Figures 2 and 3 (courtesy of Adrian P. Hunt) show that these large coprolites have a typical elongate form, with a circular cross section and up to 12 disc-shaped segments. One end is rounded, the other pointed triangular. If such objects would have been present in the Maastricht limestones, we would certainly have found them by now; that’s what I am sure of.
But why then are they not? One possible explanation is that special conditions are called for to fossilise such objects. In North America they are found in sand- and silt-rich rocks and often also enclosed in concretions (figure 3): hardened rounded to elongate nodules and cobbles in which these coprolites could easily survive. In addition, the Cretaceous sea in North America was much deeper than the one in the Maastricht area and will have had less oxygen near the sea floor. In our shallow, subtropical sea these mosasaur droppings could easily have fallen victim to the clearing actions of certain bottom-dwelling organisms; bacteria and coprophagous animals abounded.
However: that is just one view; my view. It would be good if collectors could screen their collections and come up with the goods! We can certainly exchange views and ideas via the museum website (www.nhmmaastricht.nl) on a regular basis. A word of thanks to Mart Deckers and Adrian P. Hunt for supplying photographs.

Figure 2. Mosasaur poo – the type specimen of Beechybromus wellschi Hunt & Lucas, 2018 contained in the collections of the Canadian Museum of Nature, Ottawa [NMC 40750] (see Hunt & Lucas, 2018, fig. 2C) (photograph courtesy of Adrian P. Hunt)

Figure 3. Mosasaur poo in a concretion, also referred to Beechybromus wellschi Hunt & Lucas, 2018, contained in the collections of the Canadian Museum of Nature, Ottawa [NMC 40749] (see Hunt & Lucas, 2018, fig. 3D) (photograph courtesy of Adrian P. Hunt)
Further reading
Hunt, A.P. & Lucas, S.G., 2018. Mosasaur coprolites from the Bearpaw Formation (Upper Cretaceous) of Saskatchewan, Canada. In: Lucas, S.G. & Sullivan, R.M. (eds). Fossil Record 6. New Mexico Museum of Natural History and Science Bulletin, 78, 271-275. [download freely: www.nmnaturalhistory.org/publications/publications-and-bulletins]
Hunt, A.P., Lucas, S.G., Milàn, J., Lichtig, A.J. & Jagt, J.W.M., 2015. Vertebrate coprolites from Cretaceous chalk in Europe and North America and the shark surplus paradox. In: Sullivan, R. M. & Lucas, S.G. (eds). Fossil Record 4. New Mexico Museum of Natural History and Science Bulletin, 68, 63-68. [download freely: www.nmnaturalhistory.org/publications/publications-and-bulletins]
